How to turn EU regulatory reform into a platform for industrial and clinical leadership from Spanish subsidiaries.
By Ehab Soltan
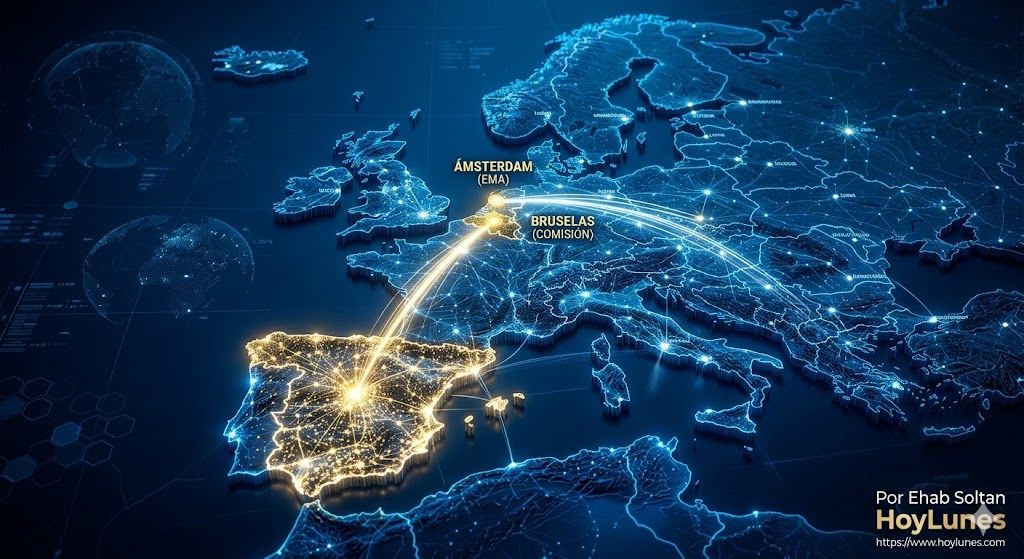
HoyLunes – Europe is redefining the rules of the pharmaceutical game. The combination of EU legislative reform, the progressive implementation of the Health Technology Assessment (HTA) Regulation, and the consolidation of the European Medicines Agency’s (EMA) scientific role is shaping a new competitive environment. In this context, the strategic question for multinationals headquartered in Spain is not defensive—how to adapt—but expansive: how to turn Spain into a leadership platform within the new European regulatory economy.
The European Union is moving toward a model that combines higher scientific standards with supranational coordination. The pharmaceutical reform package promoted by the European Commission introduces adjustments to exclusivity periods and incentives linked to equitable access. Far from being a threat, this redesign can consolidate a predictable framework for companies that know how to anticipate change.
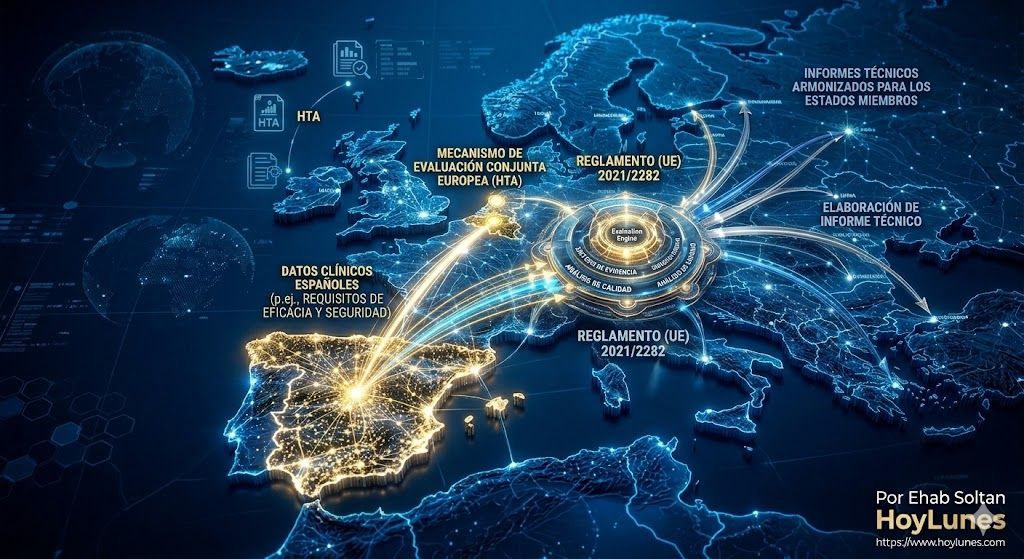
Regulation (EU) 2021/2282 introduces mandatory joint clinical assessments for certain innovative medicines. This implies that comparative evidence and added-value data can no longer be designed solely with a national logic. For Spanish subsidiaries, this means integrating the evidence generation strategy earlier into global launch planning, anticipating requirements that will simultaneously affect multiple European markets. The ability to coordinate trials, real-world evidence (RWE), and therapeutic positioning from Spain can become a strategic asset before global headquarters.
For Spanish subsidiaries, this convergence means integrating local regulatory strategy into the global planning of their parent companies, taking advantage of the fact that Spain is currently one of the EU’s main engines for high-tech product exports.
Spain as a Competitive Hub for Clinical R&D
Spain is not a peripheral player. According to the AEMPS (Spanish Agency of Medicines and Medical Devices), the country consistently ranks among European leaders in clinical trials, particularly in oncology and rare diseases. This position is the result of critical structural factors:
Highly qualified and receptive public hospital network.
Elite researchers with high scientific productivity.
Administrative agility: Spain is one of the fastest countries in the EU to launch trials thanks to Royal Decree 1090/2015.
Operational efficiency: Competitive costs compared to other major European markets.
In the current global competition—with the US accelerating approvals and Asia expanding its capacity—Spain offers a unique balance between scientific rigor and execution speed.
Sustainability as a Competitive Advantage
Pharmaceutical spending represents between 15% and 20% of health expenditure in the EU. This budgetary pressure is a reality, but also an opportunity to generate certainty. The European model, more cautious than the American one, attracts companies that prioritize scientific reputation and orderly access.
The challenge will be to lead value-based pricing and reimbursement models and managed entry agreements (risk-sharing). Those subsidiaries in Spain capable of piloting these innovative schemes will be better positioned to attract investment from their global headquarters.
Risks Requiring Corporate Leadership
The environment presents uncertainties that should not be ignored:
Adjustments to data exclusivity periods.
Regional fragmentation in market access decisions.
Growing demand for post-authorization Real-World Evidence (RWE).
Addressing these challenges with leadership turns difficulty into a competitive advantage. Companies leading in next-generation vaccines, advanced immunotherapy, or CAR-T therapies find in Spain an ideal regulatory laboratory to consolidate their internal benchmark status.

Beyond the Market: Industrial Positioning
The EU’s “open strategic autonomy” requires strengthening local production. The pharmaceutical sector in Spain is already an industrial pillar, ranking as the country’s leading sector in industrial R&D. Multinationals have the opportunity here to consolidate logistical hubs and centers of excellence that serve the entire European architecture.
According to consolidated sectoral data, the pharmaceutical industry represents the leading industrial sector in R&D investment in Spain and is one of the country’s main exporters of high-tech goods. This critical mass provides not only skilled employment and productive capacity but also the legitimacy to aspire to greater strategic responsibilities within multinational groups.
The mobilization of European funds through initiatives such as the PERTE for Vanguard Health reinforces this positioning by facilitating investments in advanced therapies, health digitalization, and critical production capacities. For multinationals, this environment reduces the initial risk of expansion and improves the medium-term return equation.
The debate is not ideological; it is structural. Will Europe be a mere consumer of external innovations or a competitive ecosystem of its own? The answer lies in the corporate decisions made today from Madrid or Barcelona.
A Strategic Invitation
HoyLunes aspires to be the platform for reflection for those who understand that innovation is not a slogan, but a shared responsibility. Multinationals with a presence in Spain have the incentive to lead the conversation on how to integrate scientific excellence, industrial competitiveness, and equitable access.
At a time when global investment committees are evaluating where to concentrate capital, talent, and regulatory capabilities, Spain’s position must not be taken for granted. Subsidiaries that take a proactive role in generating evidence, negotiating innovative access models, and industrial consolidation will be better placed to influence the strategic decisions of their parent companies.
Europe has drawn the framework. Spain has the conditions. The window of opportunity is open. The difference between consolidating leadership or losing centrality will depend on the strategic decisions adopted today in Spanish subsidiaries and defended tomorrow in global committees.
Main Sources:
European Commission – Pharmaceutical Strategy for Europe.
European Medicines Agency (EMA).
Regulation (EU) 2021/2282 (HTA).
Spanish Agency of Medicines and Medical Devices (AEMPS).
OECD Health at a Glance.
`#PharmaIndustry` `#EMA` `#ClinicalResearch` `#HoyLunes` `#EhabSoltan` `#PublicHealth`
This is for informational purposes only. For medical advice or diagnosis, consult a professional.